Mostly Cloudy: Cloud Physics for Meteorologists — Simple Classroom Demos
by Prof. Sonia Lasher-Trapp
Supercooled Water Demo
In the Midwestern U.S., we often have some really cold weather where the low overnight temperature is less than –15 °C (less than 5 °F). I like to take advantage of this weather to do this demonstration, as students are usually grumpy having to walk across campus in this cold weather anyway! The directions below are for supercooling the water outside, but you could also do this in a freezer, if you have access to one that can be undisturbed.
For this demo, I like to use the brand Fiji of bottled water. I’ve had more success with this brand because (a) the bottles are square with no ridges, allowing the students to see inside the bottle more easily, and (b) I believe it contains fewer particulates than cheaper brands. But feel free to experiment!
I usually buy 4 bottles, 2 of the smaller size and 2 of the larger size (for more drama). I put them in an out-of-the-way place not too far from the door of the building. I just place them upright within a plastic grocery bag and place a note on the top saying
“DO NOT DISTURB. SCIENCE EXPERIMENT. FOR QUESTIONS CALL PROF. LASHER-TRAPP AT (xxx) xxx-xxxx”.
This way, you won’t scare campus security that something nefarious is going on, if someone notices the bottles and call it in!
I wait until class convenes, then ask a student to go out with me and hold the door while I carry 2 of the bottles into the classroom. It’s very important that you not shake the bottles at all, or the supercooled water will freeze before you get it to the classroom!
I gently place the two bottles on 2 separate desks at the front of the class, and have students gather around. I explain how long I’ve had the bottles out there (typically 8 hours or more) and inform them of the low temperature overnight, as well as the current temperature, assuming that the latter is the current temperature of the water.
Then, I pick up one of the bottles and knock them down HARD onto the desk. Instantly the water freezes, often from the bottom toward the top of the bottle. Then I do the other one in the same way and let them observe it. (The other 2 bottles outside can be used as well, as often 1–2 bottles fail because they actually froze solid overnight, or froze as I was carrying them in.)
We then discuss why the water is able to be supercooled, but typically DOES NOT happen in a simple tray of ice cubes placed in a freezer, and why knocking the bottle on the table makes the water freeze. Occasionally something bizarre happens, perhaps depending upon the degree of supercooling and any random INP that are in the bottle. One year, a single sector plate was nucleated near the center of the bottle and grew outward (probably 5 cm across, at a slant) in time, instead of the entire bottle freezing.
During Covid, I filmed the bottles freezing at home when our class was online. Feel free to use the video below if a classroom demonstration isn’t possible! Conditions were: overnight low temperature –14 °C (6 °F), with the temperature at the time I brought the bottles inside being –9 °C (15 °F):
Our derivation says pure water can be supercooled down to –40°C = –40 °F. When we knock the bottles of supercooled water on the table, or shake it, we initiate a shock wave which helps to orient the crystals and get nucleation started at less supercooling.
You can also pour the supercooled water upon ice and it will freeze up into the bottle as you’re pouring it—see others’ videos online for this demonstration.
CCN Demo
I tend to teach during 80 minutes class periods, and this gives us a perfect amount of time to investigate what kinds of particles might make good CCN. If you teach during a shorter class period like 50 minutes, I think this will all still work out, but you’ll want everything ready to go when the students arrive.
I purchased some standard petri dishes—having about 6 is a nice number. I place enough water in the bottom of each petri dish to completely cover the bottom. Then I place some kind of little “tray” on top of the water—something flat with a rim—the tops of bottles from your collection of kitchen spices at home works well (as long they are not too tall to close the petri dish later), or the metal tops of small glass bottles of juice. I’ve also used the tops of tin cans, but you have to be careful with the sharp edges, and as you’ll see in some of the photos below, the lack of a rim around the edge means that water can seep onto its edges.
In each of these little “trays”, I multiple pieces of some kind of particle, separating each using a tweezer, so that if they deliquesce into droplets that there will be space between them. You want some particles to be soluble, and others to be insoluble, in order to explain the importance of the Kohler curves.
Some examples of partices I’ve used include:
- Big salt grains (like kosher salt)
- Small salt grains (normal table salt)
- Sugar
- Grains of sand
- Cardboard bits
- Crushed chalk
Then put all the lids of the petri dishes on. It’s best to already have spread these out among the classroom, so different “teams” can observe any evolution of their particles at various times when I inquire during the lecture (about every 15 minutes or so). Near the end of the lecture period, we talk about why the lid was needed on top the petri dish, which particles deliquesced, which did not, and why.
So simple… and yet it’s always surprising to me how much the students enjoy this activity. I think having something to break up the lecture is always a success!
Drop Sweepout Volume Demo
This is less of a “demonstration” and might be better categorized as a “learning tool”.
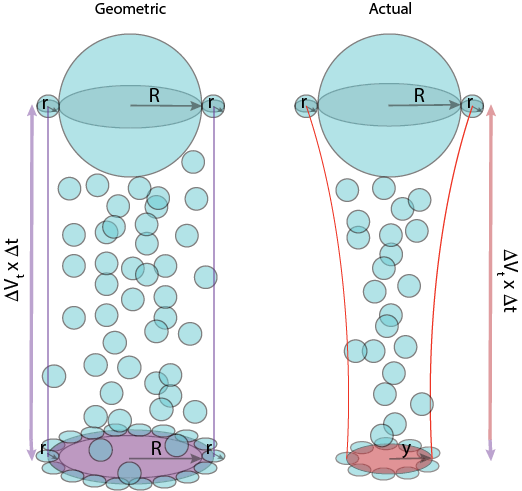
For students who find it difficult to visualize geometry in 3 dimensions, I find that using an inverted, clear tennis ball canister as the geometric sweepout volume, a tennis ball perched on top as the collector drop, and a marble/bead that I hold within or to the side of the canister (just grazing its exterior) as a cloud droplet, that has a radius that contributes to the sweepout volume (that is just wider than the canister).
In this way they can see how the size of the droplet (marble) also contributes to the sweepout volume. The depth of the can (sweepout volume) would in reality depend upon the size of the tennis ball and the marble (due to their relative fall velocities). A small droplet that does not graze the canister would not be collected (orange bead shown above).
Then you can also demonstrate how the actual droplet trajectories can bend such that a marble initially below but in the path of the falling tennis ball could actually be swept around it instead.
Seeing it this way, students that have difficulty visualizing in 3D from 2D pictures can see why the area of the sweepout volume is π (R+r) 2 :
Warm Rain Process Demo
This can be used in far less advanced classes as well as with the general public.
As most people are familiar with this experience, I just ask them to imagine a hot, humid day, when you have just taken an ice cold can of soda out of a cooler. Then they should describe what they see occur on the side of the can, and explain how this is similar to the steps of the warm rain process.
You won’t be able to demonstrate this in a classroom (if it’s that humid and hot in your classroom, no demo will make the students happy anyway!), so if you don’t have time to go outside, then you can use this video as the answer:
-
Drops are nucleated on the side of the can from soluble particles located on its surface.
-
The coldness of the can surface compared to the ambient air makes the air right over the can supersaturated , and so the drops on the can surface grow by condensation .
-
As this process continues in time, some drops are bigger than others, and they will start to slide down the sides of the can. As they do so, they bigger drops collide and coalesce with smaller drops as they “fall” along the side of the can. As they grow bigger, they slide down the can more quickly, collecting smaller drops more quickly.
-
Eventually these big drops slide off the can onto the table, the ground, etc., leaving their source of growth, as “ precipitation ”.
-
(The last possible step in the warm rain process, drop breakup, will not occur in this setup, because the can helps to keep them together during collisions.)